|
The nucleus is composed of protons and neutrons. And the energy required for transmission of a unit of information by a fiber optic cable is minuscule compared to that required to send the same information by electrical impulse over copper wire.The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. A hair-like optical fiber can transmit many times the amount of information per unit time as the thick copper wire that it replaces. The bulky wires made of relatively scarce copper formerly employed for transmitting communications signals electrically have been largely replaced by fiber optic devices consisting of transparent silica, SiO2, which transfer information as pulses of light. Furthermore, the silicon-based semiconductor devices used in solid-state electronics consumes only a fraction of the electricity once used by vacuum tube based devices. The silicon atom has 4 outer-shell electrons, half an octet, and it is a metalloid, intermediate in behavior between the metals on the left of the periodic table and the nonmetals on the right.īy vastly reducing the bulk of electronic components relative to performance, silicon has contributed to a huge saving of materials used in radios, televisions, communications equipment, and other electronic devices. Despite the value of these silicon-based products, silicon is abundant in soil and rocks, ranking second behind oxygen as a constituent of Earth’s crust. This is because silicon is the most commonly used of the semiconductor elements and during the latter 1900s provided the basis for the explosion in electronics and computers based upon semiconductor devices composed primarily of silicon. If an “element of the century” were to be named for the 1900s, humble silicon, Si, atomic number 14, atomic mass 28.09, would be a likely candidate. Furthermore, aluminum is one of the most recyclable metals, and scrap aluminum is readily melted down and cast into new aluminum goods. And there are alternative resources that can be developed, including aluminum in the fly ash left over from coal combustion. Although the ores from which aluminum is made are an extractive resource dug from the earth, aluminum is an abundant element. 
Aluminum cables also provide an efficient way to transmit electricity. So aluminum is important in energy conservation. This is because aluminum enables construction of strong lightweight components which, when used in aircraft and automobiles, require relatively less energy to move. In some important respects aluminum can be regarded as a green metal. Although it is chemically reactive, the oxide coating formed when aluminum on the surface of the metal reacts with oxygen in air is self-protecting and prevents more corrosion. Aluminum is a lightweight metal used in aircraft, automobiles, electrical transmission lines, building construction and many other applications. Magnesium is a relatively strong, very lightweight metal that is used in aircraft, extension ladders, portable tools, and other applications where light weight is particularly important.Īluminum, Al, atomic number 13, atomic mass 26.98, has 3 outer-shell electrons in addition to its 10 inner electrons. There are three isotopes of magnesium containing 12, 13, and 14 neutrons. Magnesium, Mg, atomic number 12, atomic mass 24.31, has 12 electrons per atom so it has 2 outer shell electrons. 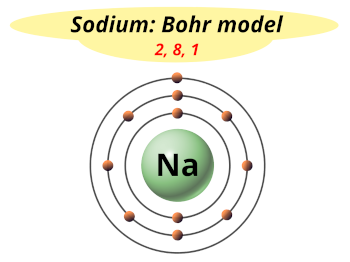
Representation of the electrons in 2 inner shells and 1 outer shell of sodium and the Lewis symbol of sodium. The electrons in sodium can be represented as shown in Figure 3.7. This is shown as a single dot in the Lewis symbol of Na in Figure 3.7. The 11th electron in the sodium atom is in a third shell, which is an outer shell. Sodium has 10 inner-shell electrons contained in its first inner shell of 2 electrons and its second one of 8 electrons. There is one major isotope of sodium containing 12 neutrons in the atom’s nucleus. Sodium, Na, atomic number 11, atomic mass 22.99, comes directly below lithium in the periodic table and is very similar to lithium in being a soft, chemically very reactive metal. The periodic table is given in Figure 3.9. Their names and properties are summarized briefly here. A total of 10 more are required to complete the 20-element abbreviated periodic table. So far in this chapter 10 elements have been defined and discussed.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
